Introduction
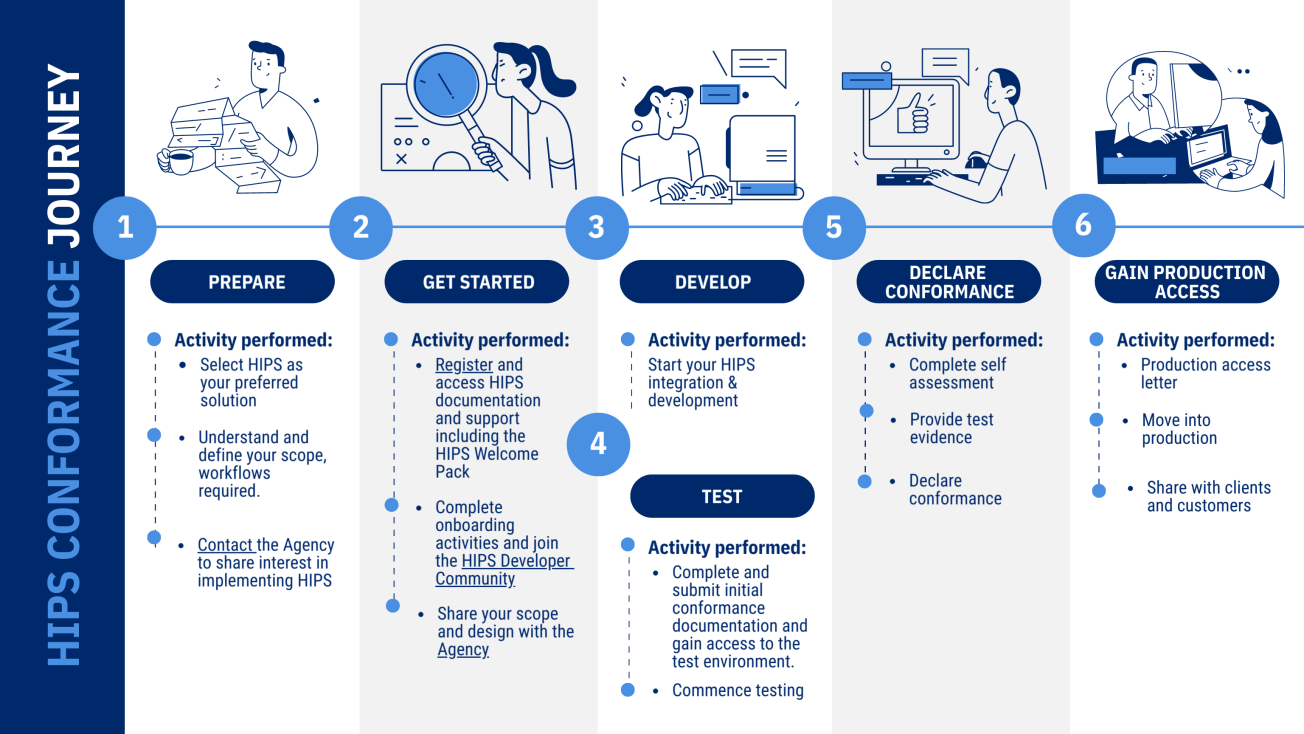
This guide will walk you through the conformance journey to integrate your hospital and laboratory systems with national digital health infrastructure through implementing Healthcare Information Provider Service (HIPS). An overview of the conformance journey for HIPS implementation using the binary code has been provided in the image below.
Pre-requisites
If you have not registered on this site, you will firstly need to “Create new account" to be able to request access to the HIPS Collaborative space.
The conformance pathway for this type of HIPS implementation requires both HI Service and My Health Record full conformance.
Overview of the HIPS Conformance Journey
Please email help@digitalhealth.gov.au if you have any questions regarding HIPS or HIPS Conformance.
To request access to the HIPS Collaborate space please email help@digitalhealth.gov.au
Once you have access to the HIPS Collaborate space, please refer to the Welcome Pack and you may like to request access to the HIPS Support Portal managed by Chamonix on behalf of the Agency.
https://collaborate.digitalhealth.gov.au/display/HIPSP/HIPS+Conformance
Step 1: Prepare
- Select HIPS as your preferred solution - visit: Healthcare Information Provider Service (HIPS) for information about HIPS
- Understand and define your scope, workflows and outcomes required. Consider timeframe, resourcing and any mandatory requirements. You may like to create a design document and workflow
- Contact the Agency to share your interest in implementing HIPS by emailing help@digitalhealth.gov.au
Step 2: Get Started
- Request access to the HIPS Collaborate space by emailing help@digitalhealth.gov.au
- Review the HIPS Welcome Pack and access relevant HIPS documentation available from the HIPS Collaborate space
- Share your scope, design and workflows with the Agency. This is usually requested prior to an onboarding virtual meeting
Step 3: Develop
- Start your HIPS integration
- Email help@digitalhealth.gov.au with any support questions
Step 4: Test
- Complete and submit initial conformance documentation which might include a Vendor Product Details form and gain access to the testing environment
- Undertake internal testing
Step 5: Declare conformance
- Complete self-assessment
- Provide test evidence. This might include observed testing sessions or the provision of XML request and response files, depending on the nature and scope of your HIPS implementation
- Declare conformance. Once you have successfully passed testing and completed in house testing, please complete and submit your Conformance and Compliance Declaration form.
Step 6: Gain Production Access
- If approved, you will receive a production access letter and be granted production access
- Move from testing into production